 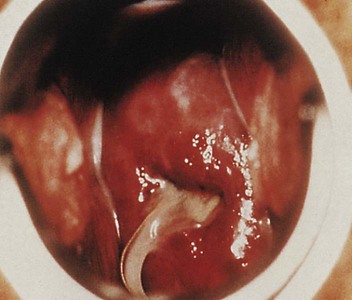
Detection of nucleic acid via nucleic acid amplification (e.g., Polymerase Chain Reaction ) OR.gonorrhoeae in a clinical specimen by detection of antigen OR A person with laboratory isolation of typical Gram-negative, oxidase-positive diplococcic by culture (presumptive Neisseria gonorrhoeae) from a clinical specimen OR.Demonstration of Gram-negative intracellular diplococci in an endocervical smear obtained from a female.Demonstration of Gram-negative intracellular diplococci in a urethral smear obtained from a male OR.gonorrhoeae in a clinical specimen by detection of antigen or nucleic acid.Ī clinically compatible case where there is: Isolation of typical Gram-negative, oxidase-positive diplococci (presumptive N.Observation of Gram-negative intracellular diplococcic in a urethral smear obtained from a male or an endocervical smear obtained from a female OR.Case Definition Clinical DescriptionĪ sexually transmitted infection commonly manifested by urethritis, cervicitis, proctitis, salpingitis, or pharyngitis. gonorrhoeae) is a Gram-negative diplococcal bacterium. Clinical: treatment name, dose, start date, and whether antibiotic resistance is suspected.Laboratory: date collected, test name, and result.  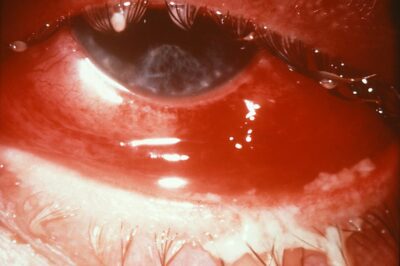
The Positive Laboratory Findings for Reportable Disease Form (HEA 3333) may be used for laboratories without access to ODRS or electronic laboratory reporting (ELR) to report positive results. For healthcare providers without access to ODRS, the Ohio Confidential Reportable Disease Form (HEA 3334) may be used. The Ohio Disease Reporting System (ODRS) should be used to report cases and lab findings to the Ohio Department of Health (ODH). If patient residence is unknown, report to the local public health department in which the reporting healthcare provider or laboratory is located. Report a case, suspected case, and/or positive laboratory result to the local public health department in which the patient resides by the close of the next business day. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
